Microbiologically induced deterioration (MID) in concrete, also referred to as microbial-induced corrosion (MIC) or concrete corrosion, is the result of acids from microbial activity degrading concrete components. MID negatively affects the structural integrity of sewer pipelines and other concrete structures (1). There are relatively few effective countermeasures to MID. In fact, even structures designed to withstand aggressive environments such as those with sulfates—concrete made with C3A-free cement, fly ash, and a water to cement ratio of 0.35—can reach a corrosion rate of 1 cm/year within a decade of installation (2).
And it is not always practical to alter the C3A (tricalcium aluminate) content of concrete. C3A content in concrete, for instance, is associated with decreased setting time, accelerated hardening, and denser pore structure (3). Depending solely on novel concrete mixtures to promote MID resistance is insufficient. In fact, the latest research recommends an integrated approach that combines modifications to concrete mixtures, concrete protective coatings, and treatments with biocides (1).
In this article, we will provide an overview of MID, discuss the concept of an integrated MID/MIC solution, and explore existing concrete protection options.
An overview of microbiologically induced deterioration/corrosion
While there are currently 22 known microorganisms that affect MID, most of our knowledge is concentrated in Thiobacillus (see Table 1 for a list of known MID microorganisms).
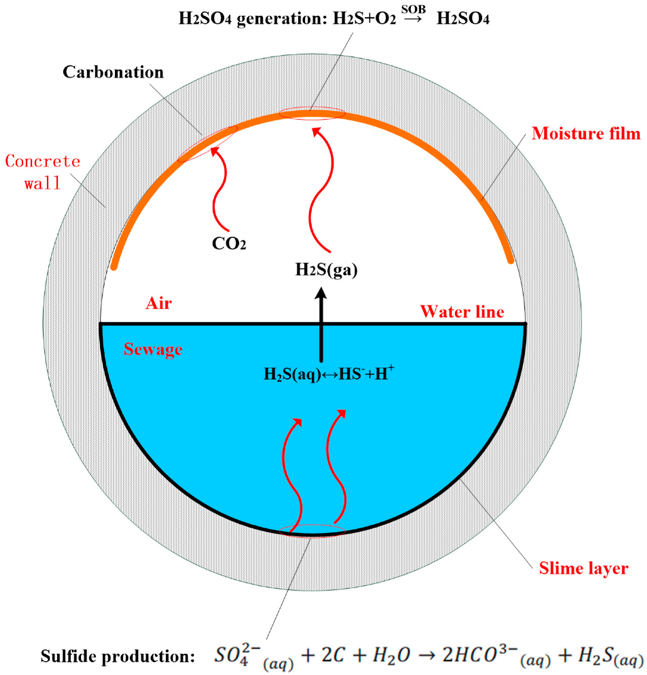
For sewer systems, the MID/MIC process begins when aqueous H2S is converted from sulfate by bacteria in a submerged slime layer. The gas form of H2S is then released and is dissolved in a moisture film (also known as a biofilm) at the tunnel’s crown. In the last step, the dissolved H2S is converted to sulfuric acid (H2SO4) by sulfur-oxidizing bacteria such as Thiobacillus (see Figure 1 for a visual schematic of the process) (4).
Mitigating the MID/MIC process depends largely on preventing bacterial adhesion and the development of biofilms. By preventing the development of biofilms, key steps in the MID/MIC process are inhibited and therefore biocorrosion can be prevented. The application of polymer coatings has been leveraged as a corrosion protection strategy against MID/MIC for decades (5). Polymer coatings, however, can suffer from poor adhesion to the base materials and can quickly degrade (6). While innovations such as conductive polymer coatings and graphene coating show significant promise, other factors such as high backpressure still pose a problem (5, 6).
Protecting concrete from MID/MIC using an integrated solution
The annual cost of corrosion is approaching US$1 trillion, with MID/MIC accounting for nearly half of the cost (6). Much of these costs are associated with frequent rehabilitation. For instance, it is possible to stop MID/MIC through regular high-pressure washing of the concrete surface, which removes the biofilm. However, it takes between 60 and 140 days for the bacteria to recover the biofilm to prewash levels (5).
Instead of focusing on temporary answers, a better approach relies on combining concrete mixtures, chemical intervention, and protective coatings into an integrated solution. The solution takes a multi-pronged approach to inhibit biofilm growth and prevent concrete biodeterioration (1).
The first layer defense is altering the concrete composition to decrease the rate of corrosion should initial biofilm inhibiting efforts fail. Decreasing the rate of corrosion is a mitigating factor that helps reduce potential rehabilitation costs. Effective mixtures include incorporating silica fume concrete, polymer-modified mortar, and C3A. These mixtures can increase the strength, durability, and pore density of concrete structures to lower the rate of corrosion (1). It is also possible to incorporate antimicrobial aggregates within the concrete mixture through low-cost sorbents and metal solutions such as copper and copper/cobalt (7).
The second layer of defense uses chemical intervention to control MIC by raising the pH of the sewage (e.g., magnesium hydroxide), inhibiting growth (biocides e.g., sodium bromide, sodium hydroxide), or interrupting the process by oxidizing sulfide directly (e.g., hydrogen peroxide) (4).
The last layer of defense, which was described earlier, uses a protective coating to inhibit biofilm growth (5).
In systems where known MID/MIC microorganisms are present, these layers of defense are very effective in slowing down the development of biofilms and preventing concrete corrosion. However, the full implementation of this integrated solution is not always possible.
Chemical intervention, for instance, carries a significant, recurring cost. Additionally, chemical intervention can lead to environmental problems as a result of unintended chemical reactions. For existing systems, replacing concrete structures with concrete using enhanced mixtures is unlikely. And protective coatings have an even shorter service life in concrete systems with high backpressure. Using a concrete waterproofing membrane—also known as a concrete protective liner (CPL)—in place of a polymer coating offers an alternative approach.
Incorporating CPL in an integrated solution for microbiologically induced deterioration
A CPL offers many of the same benefits of polymer coatings such as low permeability and high chemical resistance, with the added benefit of enhanced durability and strength. By being structurally attached to concrete surfaces, CPL achieves significantly longer service life when compared with spray-on concrete coatings.
Develop an integrated solution to microbiologically induced deterioration in concrete by combining novel concrete mixtures, chemical intervention techniques, and concrete protective liners.
Searching for the right concrete protective liner for your integrated solution to MID/MIC? AGRU-ULTRA GRIP® is a CPL made with HDPE, HDPE-el, or PP and offers unparalleled backpressure resistance through a revolutionary redesign of AGRU’s patented Sure-Grip anchor system. Ultra Grip has a pullout resistance of up to 820 kN/m2 with PE 80 at 20°C in 3,000 psi concrete. Ultra Grip’s distinctive V-shape anchor system can resist long-term sustained backpressure of up to 1.75 bar (at 20°C or 68°F). Reach out to AGRU today to learn more about Ultra Grip and its role in an integrated solution to microbiologically induced deterioration in concrete.
Contact AGRU
References
- 1. Wei et al., “Microbiologically induced deterioration of concrete – A Review.” Braz. J. Microbiol. 44(4): 1001–1007. (2013). Accessed online 13 November 2019 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3958164/.
- 2. Grengg et al., “Microbiologically induced concrete corrosion: A case study from a combined sewer network.” Cement and Concrete Res. 77: 16-25. (2015). Accessed online 13 November 2019. https://www.sciencedirect.com/science/article/pii/S0008884615001805.
- 3. J. Kim et al., “The Influence of C3A Content in Cement on the Chloride Transport.” Adv. Mat. Sci. Eng. Vol. 2016, Article ID 5962821 (2016). Accessed online 13 November 2019. https://www.hindawi.com/journals/amse/2016/5962821/.
- 4. Wu et al., “The Sustainability of Concrete in Sewer Tunnel—A Narrative Review of Acid Corrosion in the City of Edmonton, Canada.” Sustainability. 10(2): 517. (2018). https://www.mdpi.com/2071-1050/10/2/517/htm.
- 5. Guo et al., “Polymers for Combating Biocorrosion.” Front. Mater. (2018). Accessed online 13 November 2019. https://www.frontiersin.org/articles/10.3389/fmats.2018.00010/full.
- 6. Krishnamurthy et al., “Superiority of Graphene over Polymer Coatings for Prevention of Microbially Induced Corrosion.” Sci. Reports. 5, 13858. (2015). Accessed online 13 November 2019. https://www.nature.com/articles/srep13858.
- 7. Caicedo-Ramirez, “Antimicrobial Aggregates for the In-Situ Control of Microbially Induced Concrete Corrosion.” University of Colorado, Boulder. Civil Engineering Graduate Theses & Dissertations. 350. (2018). Accessed online 13 November 2019. https://scholar.colorado.edu/cven_gradetds/350.
Table 1. Organisms involved in the deterioration of concrete. From reference 1.
| Bacteria | |
| Thiobacillus intermedius | Giannantonio et al., 2009; Magniont et al., 2011 |
| Thiobacillus neapolitanus | Magniont et al., 2011; Mustafa, 2009 |
| Thiobacillus novellus | Magniont et al., 2011; Mustafa, 2009 |
| Thiobacillus thioparus | Magniont et al., 2011; Mustafa, 2009 |
| Acidithiobacillus thiooxidans | Lahav et al., 2004; Magniont et al., 2011; Mustafa, 2009; Vollertsen et al., 2008 |
| Thiomonas perometablis | Vollertsen et al., 2008 |
| Fungus | |
| Alternaria sp | Ghafoori and Mathis, 1997 |
| Cladosporium cladosporioides | Ghafoori and Mathis, 1997 |
| Epicoccum nigrum | Ghafoori and Mathis, 1997 |
| Fusarium sp | Ghafoori and Mathis, 1997; Giannantonio et al., 2009 |
| Mucor sp | Ghafoori and Mathis, 1997 |
| Penicillium oxalicum | Ghafoori and Mathis, 1997 |
| Pestalotiopsis maculans | Ghafoori and Mathis, 1997 |
| Trichoderma asperellum | Ghafoori and Mathis, 1997 |
| Aspergillus niger | Lajili et al., 2008 |
| Alternaria alternata | Warscheida and Braamsb, 2000 |
| Exophiala sp. | Warscheida and Braamsb, 2000 |
| Coniosporium uncinatum | Warscheida and Braamsb, 2000 |
| Algae | |
| Chaetomorpha antennina | Javaherdashti and Setareh, 2006 |
| Ulva fasciata | Javaherdashti and Setareh, 2006 |
| Lichen | |
| Acarospora cervina | Edwards et al., 1999 |
| Candelariella ssp | Edwards et al., 1999 |
Figure 1. Schematic of the microbially induced corrosion (MIC) process within a sewer. From reference 4.